Hold down the T key for 3 seconds to activate the audio accessibility mode, at which point you can click the K key to pause and resume audio. Useful for the Check Your Understanding and See Answers.
On the previous page, we learned what heat does to an object when it is gained or released. Heat gains or losses result in changes in temperature, changes in state or the performance of work. Heat is a transfer of energy. When gained or lost by an object, there will be corresponding energy changes within that object. A change in temperature is associated with changes in the average kinetic energy of the particles within the object. A change in state is associated with changes in the internal potential energy possessed by the object. And when work is done, there is an overall transfer of energy to the object upon which the work is done. In this part of Lesson 2, we will investigate the question How does one measure the quantity of heat gained or released by an object?
Specific Heat Capacity
Suppose that several objects composed of different materials are heated in the same manner. Will the objects warm up at equal rates? The answer: most likely not. Different materials would warm up at different rates because each material has its own specific heat capacity. The specific heat capacity refers to the amount of heat required to cause a unit of mass (say a gram or a kilogram) to change its temperature by 1°C. Specific heat capacities of various materials are often listed in textbooks. Standard metric units are Joules/kilogram/Kelvin (J/kg/K). More commonly used units are J/g/°C. Use the widget below to view specific heat capacities of various materials. Simply type in the name of a substance (aluminum, iron, copper, water, methanol, wood, etc.) and click on the Submit button; results will be displayed in a separate window.
The specific heat capacity of solid aluminum (0.904 J/g/°C) is different than the specific heat capacity of solid iron (0.449 J/g/°C). This means that it would require more heat to increase the temperature of a given mass of aluminum by 1°C compared to the amount of heat required to increase the temperature of the same mass of iron by 1°C. In fact, it would take about twice as much heat to increase the temperature of a sample of aluminum a given amount compared to the same temperature change of the same amount of iron. This is because the specific heat capacity of aluminum is nearly twice the value of iron.
Heat capacities are listed on a per gram or per kilogram basis. Occasionally, the value is listed on a per mole  basis, in which case it is called the molar heat capacity. The fact that they are listed on a per amount basis is an indication that the quantity of heat required to raise the temperature of a substance depends on how much substance there is. Any person who has boiled a pot of water on a stove, undoubtedly know this truth. Water boils at 100°C at sea level and at slightly lowered temperatures at higher elevations. To bring a pot of water to a boil, its temperature must first be raised to 100°C. This temperature change is achieved by the absorption of heat from the stove burner. One quickly notices that it takes considerably more time to bring a full pot of water to a boil than to bring a half-full of water to a boil. This is because the full pot of water must absorb more heat to result in the same temperature change. In fact, it requires twice as much heat to cause the same temperature change in twice the mass of water.
basis, in which case it is called the molar heat capacity. The fact that they are listed on a per amount basis is an indication that the quantity of heat required to raise the temperature of a substance depends on how much substance there is. Any person who has boiled a pot of water on a stove, undoubtedly know this truth. Water boils at 100°C at sea level and at slightly lowered temperatures at higher elevations. To bring a pot of water to a boil, its temperature must first be raised to 100°C. This temperature change is achieved by the absorption of heat from the stove burner. One quickly notices that it takes considerably more time to bring a full pot of water to a boil than to bring a half-full of water to a boil. This is because the full pot of water must absorb more heat to result in the same temperature change. In fact, it requires twice as much heat to cause the same temperature change in twice the mass of water.
Specific heat capacities are also listed on a per K or a per °C basis. The fact that the specific heat capacity is listed on a per degree basis is an indication that the quantity of heat required to raise a given mass of substance to a specific temperature depends upon the change in temperature required to reach that final temperature. In other words, it is not the final temperature that is of importance, it is the overall temperature change. It takes more heat to change the temperature of water from 20°C to 100°C (a change of 80°C) than to increase the temperature of the same amount of water from 60°C to 100°C (a change of 40°C). In fact, it requires twice as much heat to change the temperature of a given mass of water by 80°C compared to the change of 40°C. A person who wishes to bring water to a boil on a stovetop more quickly should begin with warm tap water instead of cold tap water.
This discussion of specific heat capacity deserves one final comment. The term specific heat capacity is somewhat of a misnomer. The term implies that substances may have the ability to contain a thing called heat. As has been previously discussed, heat is not something that is contained in an object. Heat is something that is transferred to or from an object. Objects contain energy in a variety of forms. When that energy is transferred to other objects of different temperatures, we refer to transferred energy as heat or thermal energy. While it's not likely to catch on, a more appropriate term would be specific energy capacity.
Relating the Quantity of Heat to the Temperature Change
Specific heat capacities provide a means of mathematically relating the amount of thermal energy gained (or lost) by a sample of any substance to the sample's mass and its resulting temperature change. The relationship between these four quantities is often expressed by the following equation.
Q = m•C•ΔT
 where Q is the quantity of heat transferred to or from the object, m is the mass of the object, C is the specific heat capacity of the material the object is composed of, and ΔT is the resulting temperature change of the object. As in all situations in science, a delta (∆) value for any quantity is calculated by subtracting the initial value of the quantity from the final value of the quantity. In this case, ΔT is equal to Tfinal - Tinitial. When using the above equation, the Q value can turn out to be either positive or negative. As always, a positive and a negative result from a calculation has physical significance. A positive Q value indicates that the object gained thermal energy from its surroundings; this would correspond to an increase in temperature and a positive ΔT value. A negative Q value indicates that the object released thermal energy to its surroundings; this would correspond to a decrease in temperature and a negative ΔT value.
where Q is the quantity of heat transferred to or from the object, m is the mass of the object, C is the specific heat capacity of the material the object is composed of, and ΔT is the resulting temperature change of the object. As in all situations in science, a delta (∆) value for any quantity is calculated by subtracting the initial value of the quantity from the final value of the quantity. In this case, ΔT is equal to Tfinal - Tinitial. When using the above equation, the Q value can turn out to be either positive or negative. As always, a positive and a negative result from a calculation has physical significance. A positive Q value indicates that the object gained thermal energy from its surroundings; this would correspond to an increase in temperature and a positive ΔT value. A negative Q value indicates that the object released thermal energy to its surroundings; this would correspond to a decrease in temperature and a negative ΔT value.
Knowing any three of these four quantities allows an individual to calculate the fourth quantity. A common task in many physics classes involves solving problems associated with the relationships between these four quantities. As examples, consider the two problems below. The solution to each problem is worked out for you. Additional practice can be found in the Check Your Understanding section at the bottom of the page.
|
Example Problem 1
What quantity of heat is required to raise the temperature of 450 grams of water from 15°C to 85°C? The specific heat capacity of water is 4.18 J/g/°C.
|
Like any problem in physics, the solution begins by identifying known quantities and relating them to the symbols used in the relevant equation. In this problem, we know the following:
m = 450 g
C = 4.18 J/g/°C
Tinitial = 15°C
Tfinal = 85°C
We wish to determine the value of Q - the quantity of heat. To do so, we would use the equation Q = m•C•ΔT. The m and the C are known; the ΔT can be determined from the initial and final temperature.
T = Tfinal - Tinitial = 85°C - 15°C = 70.°C
With three of the four quantities of the relevant equation known, we can substitute and solve for Q.
Q = m•C•ΔT = (450 g)•(4.18 J/g/°C)•(70.°C)
Q = 131670 J
Q = 1.3x105 J = 130 kJ (rounded to two significant digits)
|
Example Problem 2
A 12.9 gram sample of an unknown metal at 26.5°C is placed in a Styrofoam cup containing 50.0 grams of water at 88.6°C. The water cools down and the metal warms up until thermal equilibrium is achieved at 87.1°C. Assuming all the heat lost by the water is gained by the metal and that the cup is perfectly insulated, determine the specific heat capacity of the unknown metal. The specific heat capacity of water is 4.18 J/g/°C.
|
Compared to the previous problem, this is a much more difficult problem. In fact, this problem is like two problems in one. At the center of the problem-solving strategy is the recognition that the quantity of heat lost by the water (Qwater) equals the quantity of heat gained by the metal (Qmetal). Since the m, C and ΔT values of the water are known, the Qwater can be calculated. This Qwater value equals the Qmetal value. Once the Qmetal value is known, it can be used with the m and ΔT value of the metal to calculate the Qmetal. Use of this strategy leads to the following solution:
Part 1: Determine the Heat Lost by the Water
Given:
m = 50.0 g
C = 4.18 J/g/°C
Tinitial = 88.6°C
Tfinal = 87.1°C
ΔT = -1.5°C (Tfinal - Tinitial)
Solve for Qwater:
Qwater = m•C•ΔT = (50.0 g)•(4.18 J/g/°C)•(-1.5°C)
Qwater = -313.5 J (unrounded)
(The - sign indicates that heat is lost by the water)
Part 2: Determine the value of Cmetal
Given:
Qmetal = 313.5 J (use a + sign since the metal is gaining heat)
m = 12.9 g
Tinitial = 26.5°C
Tfinal = 87.1°C
ΔT = (Tfinal - Tinitial )
Solve for Cmetal:
Rearrange Qmetal = mmetal•Cmetal•ΔTmetal to obtain Cmetal = Qmetal / (mmetal•ΔTmetal)
Cmetal = Qmetal / (mmetal•ΔTmetal) = (313.5 J)/[(12.9 g)•(60.6°C)]
Cmetal = 0.40103 J/g/°C
Cmetal = 0.40 J/g/°C (rounded to two significant digits)
Heat and Changes of State
The discussion above and the accompanying equation (Q = m•C•∆T) relates the heat gained or lost by an object to the resulting temperature changes of that object. As we have learned, sometimes heat is gained or lost but there is no temperature change. This is the case when the substance is undergoing a state change. So now we must investigate the mathematics related to changes in state and the quantity of heat.
To begin the discussion, let's consider the various state changes that could be observed for a sample of matter. The table below lists several state changes and identifies the name commonly associated with each process.
|
Process
|
Change of State
|
|
Melting
|
Solid to Liquid
|
|
Freezing
|
Liquid to Solid
|
|
Vaporization
|
Liquid to Gas
|
|
Condensation
|
Gas to Liquid
|
|
Sublimation
|
Solid to Gas
|
|
Deposition
|
Gas to Solid
|
In the case of melting, boiling and sublimation, energy would have to be added to the sample of matter in order to cause the change of state. Such state changes are referred to as being endothermic. Freezing, condensation and deposition are exothermic; energy is released by the sample of matter when these state changes occur. So one might notice that a sample of ice (solid water) undergoes melting when it is placed on or near a burner. Heat is transferred from the burner to the sample of ice; energy is gained by the ice causing the change of state. But how much energy would be required to cause such a change of state? Is there a mathematical formula that might help in determining the answer to this question? There most certainly is.
The amount of energy required to change the state of a sample of matter depends on three things. It depends upon what the substance is, on how much substance is undergoing the state change, and upon what state change that is occurring. For instance, it requires a different amount of energy to melt ice (solid water) compared to melting iron. And it requires a different amount of energy to melt ice (solid water) as it does to vaporize the same amount of liquid water. And finally, it requires a different amount of energy to melt 10.0 grams of ice compared to melting 100.0 grams of ice. The substance, the process and the amount of substance are the three variables that affect the amount of energy required to cause a specific change in state. Use the widget below to investigate the effect of the substance and the process upon the energy change. (Note that the Heat of Fusion is the energy change associated with the solid-liquid state change.)
The values for the specific heat of fusion and the specific heat of vaporization are reported on a per amount basis. For instance, the specific heat of fusion of water is 333 J/gram. It takes 333 J of energy to melt 1.0 gram of ice. It takes 10 times as much energy - 3330 J - to melt 10.0 grams of ice. Reasoning in this manner leads to the following formulae relating the quantity of heat to the mass of the substance and the heat of fusion and vaporization.
For melting and freezing: Q = m•ΔHfusion
For vaporization and condensation: Q = m•ΔHvaporization
where Q represents the quantity of energy gained or released during the process, m represents the mass of the sample, ΔHfusion represents the specific heat of fusion (on a per gram basis) and ΔHvaporization represents the specific heat of vaporization (on a per gram basis). Similar to the discussion regarding Q = m•C•ΔT, the values of Q can be either positive or negative. Values of Q are positive for the melting and vaporization process; this is consistent with the fact that the sample of matter must gain energy in order to melt or vaporize. Values of Q are negative for the freezing and condensation process; this is consistent with the fact that the sample of matter must lose energy in order to freeze or condense.
As an illustration of how these equations can be used, consider the following two example problems.
|
Example Problem 3
Elise places 48.2 grams of ice in her beverage. What quantity of energy would be absorbed by the ice (and released by the beverage) during the melting process? The heat of fusion of water is 333 J/g.
|
The equation relating the mass (48.2 grams), the heat of fusion (333 J/g), and the quantity of energy (Q) is Q = m•ΔHfusion. Substitution of known values into the equation leads to the answer.
Q = m•ΔHfusion = (48.2 g)•(333 J/g)
Q = 16050.6 J
Q = 1.61 x 104 J = 16.1 kJ (rounded to three significant digits)
Example Problem 3 involves a rather straightforward, plug-and-chug type calculation. Now we will try Example Problem 4, which will require a significant deeper level of analysis.
|
Example Problem 4
What is the minimum amount of liquid water at 26.5 degrees that would be required to completely melt 50.0 grams of ice? The specific heat capacity of liquid water is 4.18 J/g/°C and the specific heat of fusion of ice is 333 J/g.
|
In this problem, the ice is melting and the liquid water is cooling down. Energy is being transferred from the liquid to the solid. To melt the solid ice, 333 J of energy must be transferred for every gram of ice. This transfer of energy from the liquid water to the ice will cool the liquid down. But the liquid can only cool as low as 0°C - the freezing point of the water. At this temperature the liquid will begin to solidify (freeze) and the ice will not completely melt.
We know the following about the ice and the liquid water:
Given Info about Ice:
m = 50.0 g
ΔHfusion = 333 J/g
Given Info about Liquid Water:
C = 4.18 J/g/°C
Tinitial = 26.5°C
Tfinal = 0.0°C
ΔT = -26.5°C (Tfinal - Tinitial )
The energy gained by the ice is equal to the energy lost from the water.
Qice = -Qliquid water
The - sign indicates that the one object gains energy and the other object loses energy. We can calculate the left side of the above equation as follows:
Qice = m•ΔHfusion = (50.0 g)•(333 J/g)
Qice = 16650 J
Now we can set the right side of the equation equal to m•C•ΔT and begin to substitute in known values of C and ΔT in order to solve for the mass of the liquid water. The solution is:
16650 J = -Qliquid water
16650 J = -mliquid water•Cliquid water•ΔTliquid water
16650 J = -mliquid water•(4.18 J/g/°C)•(-26.5°C)
16650 J = -mliquid water•(-110.77 J/°C)
mliquid water = -(16650 J)/(-110.77 J/°C)
mliquid water = 150.311 g
mliquid water = 1.50x102 g (rounded to three significant digits)
Heating and Cooling Curves Revisited
On the previous page of Lesson 2, the heating curve of water was discussed. The heating curve showed how the temperature of water increased over the course of time as a sample of water in its solid state (i.e., ice) was heated. We learned that the addition of heat to the sample of water could cause either changes in temperature or changes in state. At the melting point of water, the addition of heat causes a transformation of the water from the solid state to the liquid state. And at the boiling point of water, the addition of heat causes a transformation of the water from the liquid state to the gaseous state. These changes in state occurred without any changes in temperature. However, the addition of heat to a sample of water that is not at any phase change temperatures will result in a change in temperature.
Now we can approach the topic of heating curves on a more quantitative basis. The diagram below represents the heating curve of water. There are five labeled sections on the plotted lines.
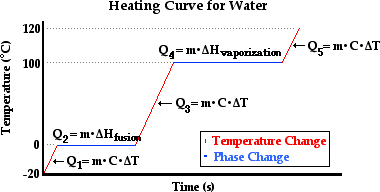
The three diagonal sections represent the changes in temperature of the sample of water in the solid state (section 1), the liquid state (section 3), and the gaseous state (section 5). The two horizontal sections represent the changes in state of the water. In section 2, the sample of water is undergoing melting; the solid is changing to a liquid. In section 4, the sample of water is undergoing boiling; the liquid is changing to a gas. The quantity of heat transferred to the water in sections 1, 3, and 5 is related to the mass of the sample and the temperature change by the formula Q = m•C•ΔT. And the quantity of heat transferred to the water in sections 2 and 4 is related to the mass of the sample and the heat of fusion and vaporization by the formulae Q = m•ΔHfusion (section 2) and Q = m•ΔHvaporization (section 4). So now we will make an effort to calculate the quantity of heat required to change 50.0 grams of water from the solid state at -20.0°C to the gaseous state at 120.0°C. The calculation will require five steps - one step for each section of the above graph. While the specific heat capacity of a substance varies with temperature, we will use the following values of specific heat in our calculations:
Solid Water: C=2.00 J/g/°C
Liquid Water: C = 4.18 J/g/°C
Gaseous Water: C = 2.01 J/g/°C
Finally, we will use the previously reported values of ΔHfusion (333 J/g) and ΔHvaporization (2.23 kJ/g).
Section 1: Changing the temperature of solid water (ice) from -20.0°C to 0.0°C.
Use Q1 = m•C•ΔT
where m = 50.0 g, C = 2.00 J/g/°C, Tinitial = -200°C, andTfinal = 0.0°C
Q1 = m•C•ΔT = (50.0 g)•(2.00 J/g/°C)•(0.0°C - -20.0°C)
Q1 = 2.00 x103 J = 2.00 kJ
Section 2: Melting the Ice at 0.0°C.
Use Q2 = m•ΔHfusion
where m = 50.0 g and ΔHfusion = 333 J/g
Q2 = m•ΔHfusion = (50.0 g)•(333 J/g)
Q2 = 1.665 x104 J = 16.65 kJ
Q2 = 16.7 kJ (rounded to 3 significant digits)
Section 3: Changing the temperature of liquid water from 0.0°C to 100.0°C.
Use Q3 = m•C•ΔT
where m = 50.0 g, C = 4.18 J/g/°C, Tinitial = 0.0°C, and Tfinal = 100.0°C
Q3 = m•C•ΔT = (50.0 g)•(4.18 J/g/°C)•(100.0°C - 0.0°C)
Q3 = 2.09 x104 J = 20.9 kJ
Section 4: Boiling the Water at 100.0°C.
Use Q4 = m•ΔHvaporization
where m = 50.0 g and ΔHvaporization = 2.23 kJ/g
Q4 = m•ΔHvaporization = (50.0 g)•(2.23 kJ/g)
Q4 = 111.5 kJ
Q4 = 112 kJ (rounded to 3 significant digits)
Section 5: Changing the temperature of liquid water from 100.0°C to 120.0°C.
Use Q5 = m•C•ΔT
where m = 50.0 g, C = 2.01 J/g/°C, Tinitial = 100.0°C, and Tfinal = 120.0°C
Q5 = m•C•ΔT = (50.0 g)•(2.01 J/g/°C)•(120.0°C - 100.0°C)
Q5 = 2.01 x103 J = 2.01 kJ
The total amount of heat required to change solid water (ice) at -20°C to gaseous water at 120°C is the sum of the Q values for each section of the graph. That is,
Qtotal = Q1 + Q2 + Q3 + Q4 + Q5
Summing these five Q values and rounding to the proper number of significant digits leads to a value of 154 kJ as the answer to the original question.
In the above example, there are several features of the solution that are worth reflecting on:
- First: The lengthy problem was divided into parts, with each part representing one of the five sections of the graph. Since there were five Q values being calculated, they were labeled as Q1, Q2, etc. This level of organization is required in a multi-step problem such as this one.
- Second: Attention was given to the +/- sign on ΔT. The change in temperature (or of any quantity) is always calculated as the final value of the quantity minus the initial value of that quantity.
- Third: Attention was given to units throughout the course of the problem. Units of Q will either be in Joule or kiloJoule depending on which quantities are being multiplied. Failure to pay attention to units is a common cause of failure in problems like these.
- Fourth: Attention was given to significant digits throughout the course of the problem. While this should never become the major emphasis of any problem in physics, it is certainly a detail worth attending to.
We've learned here on this page how to calculate the quantity of heat involved in any heating/cooling process and in any change of state process. This understanding will be critical as we proceed to the next page of Lesson 2 on the topic of calorimetry. Calorimetry is the science associated with determining the changes in energy of a system by measuring the heat exchanged with the surroundings.
Check Your Understanding
1. Water has an unusually high specific heat capacity. Which one of the following statements logically follows from this fact?
a. Compared to other substances, hot water causes severe burns because it is a good conductor of heat.
b. Compared to other substances, water will quickly warm up to high temperatures when heated.
c. Compared to other substances, it takes a considerable amount of heat for a sample of water to change its temperature by a small amount.
2. Explain why large bodies of water such as Lake Michigan can be quite chilly in early July despite the outdoor air temperatures being near or above 90°F (32°C).
3. The table below describes a thermal process for a variety of objects (indicated by red, bold-faced text). For each description, indicate if heat is gained or lost by the object, whether the process is endothermic or exothermic, and whether Q for the indicated object is a positive or negative value.
| |
Process |
Heat Gained or Heat Lost?
|
Endo- or Exothermic?
|
Q: + or -?
|
|
a.
|
An ice cube is placed into a glass of room temperature lemonade in order to cool the beverage down. |
|
|
|
|
b.
|
A cold glass of lemonade sits on the picnic table in the hot afternoon sun and warms up to 32°F. |
|
|
|
|
c.
|
The burners on an electric stove are turned off and gradually cool down to room temperature. |
|
|
|
|
d.
|
The teacher removes a large chunk of dry ice from a thermos and places it into water. The dry ice sublimes, producing gaseous carbon dioxide. |
|
|
|
|
e.
|
Water vapor in the humidified air strikes the window and turns to a dew drop (drop of liquid water). |
|
|
|
4. An 11.98-gram sample of zinc metal is placed in a hot water bath and warmed to 78.4°C. It is then removed and placed into a Styrofoam cup containing 50.0 mL of room temperature water (T=27.0°C; density = 1.00 g/mL). The water warms to a temperature of 28.1°C. Determine the specific heat capacity of the zinc.
5. Jake grabs a can of soda from the closet and pours it over ice in a cup. Determine the amount of heat lost by the room temperature soda as it melts 61.9 g of ice (ΔHfusion = 333 J/g).
6. The heat of sublimation (ΔHsublimation) of dry ice (solid carbon dioxide) is 570 J/g. Determine the amount of heat required to turn a 5.0-pound bag of dry ice into gaseous carbon dioxide. (Given: 1.00 kg = 2.20 lb)
7. Determine the amount of heat required to increase the temperature of a 3.82-gram sample of solid para-dichlorobenzene from 24°C to its liquid state at 75°C. Para-dichlorobenzene has a melting point of 54°C, a heat of fusion of 124 J/g and specific heat capacities of 1.01 J/g/°C (solid state) and 1.19 J/g/°C (liquid state).